|
# # # # This week the pharmaceutical company TEVA Pharmaceuticals Industries Ltd has announced a deal with a small German biotech firm called MODAG. The two companies are forming a strategic collaboration on the exclusive worldwide licensing and development of MODAG’s lead compound anle138b. Anle138b is a small molecule inhibitor of the believed to be toxic forms of the Parkinson’s-associated protein alpha synuclein. In today’s post, we will discuss what is known about anle138b and the implications of this new partnership. # # # # |

In 1901, Haim Salomon and his brother-in-law Moshe Levin established a small wholesale drug business, near the Nablus Gate in Jerusalem. They called it “Salomon and Levin”. A few years later, Yitzhak Elstein, another of Haim Salomon’s brothers-in-law, joined the firm and they changed the name of the company to SLE – Salomon Levin and Elstein.

From these humble beginning, grew a pharmaceutical juggernaut that we know today as TEVA Pharmaceuticals.
TEVA – meaning “Nature” in Hebrew – is now an international producer of pharmaceutical agents, with 40,000 employees working across 65 manufacturing facilities in more than 30 countries. The company has a portfolio of more than 3,500 medicines, and they produce approximately 85 billion tablets and capsules per year (Source).
Does TEVA produce any drugs for Parkinson’s?
Yes, Azilect (rasagiline) – an approved monoamine-oxidase B inhibitor for the treatment of Parkinson’s – was developed by Teva Pharmaceuticals.
In addition, they are actively developing novel therapies. And this week they signed a really interesting deal to collaborate with a small German biotech company called MODAG.
What does MODAG do?
MODAG has been developing an alpha synuclein aggregation inhibitor called Anle138b
And what does Anle138b do?
Anle138b is a diphenyl–pyrazole compound (don’t worry about what that means), which displays potent anti-protein aggregating properties.

In particular, this compound has been shown to inhibit the formation of the oligmeric and fribrillar forms of alpha synuclein.
Remind me one more time what is alpha synuclein?
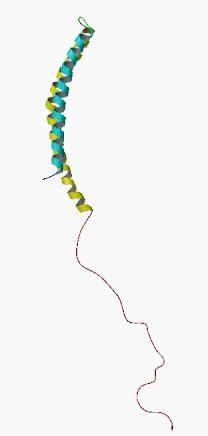
Alpha synuclein sounds like a distant galaxy, but it is one of the most common proteins in our brains. It makes up about 1% of all the protein in a neuron. When alpha synuclein protein is produced by a cell, it normally referred as a ‘natively unfolded protein’, in that is does not really have a defined structure.
When it is first produced, alpha synuclein will look something like this:
Alpha synuclein. Source: Wikipedia
In this form, alpha synuclein is considered a monomer – which is a single molecule that can bind to other molecules. When it does bind to other alpha synuclein proteins, they form an oligomer (a collection of a certain number of monomers in a specific structure). It is believed that alpha synuclein has certain functions as a monomer, but may also have specific tasks as an oligomer.
In Parkinson’s, alpha synuclein will also misfold and aggregate together to form amyloid fibrils.
Microscopic images of monomers, oligomers and fibrils. Source: Brain
And it is believed that the oligomer and fibril forms of alpha synuclein protein that aggregate together, and then go on to form what we call Lewy bodies.
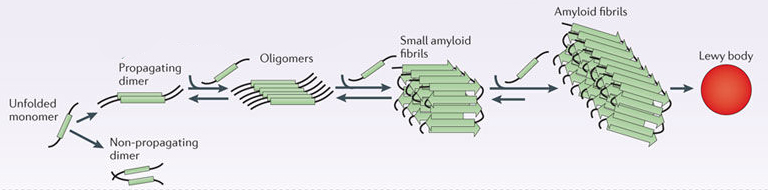
A Lewy body is referred to as a cellular inclusion, as they are almost always found inside the cell body. They are a characterisitic feature of the Parkinsonian brain.

Right, so Anle138b inhibits the oligomer and fibril forms of alpha synuclein?
Yes.
In 2013, researchers behind the company published this report:
Title: Anle138b: a novel oligomer modulator for disease-modifying therapy of neurodegenerative diseases such as prion and Parkinson’s disease.
Authors: Wagner J, Ryazanov S, Leonov A, Levin J, Shi S, Schmidt F, Prix C, Pan-Montojo F, Bertsch U, Mitteregger-Kretzschmar G, Geissen M, Eiden M, Leidel F, Hirschberger T, Deeg AA, Krauth JJ, Zinth W, Tavan P, Pilger J, Zweckstetter M, Frank T, Bähr M, Weishaupt JH, Uhr M, Urlaub H, Teichmann U, Samwer M, Bötzel K, Groschup M, Kretzschmar H, Griesinger C, Giese A.
Journal: Acta Neuropathol. 2013 Jun;125(6):795-813
PMID: 23604588 (This article is OPEN ACCESS if you would like to read it)
In this first study, the researchers discovered Anle138b by conducted a large screening study to identify for molecules that could inhibit the toxic form of alpha synuclein.
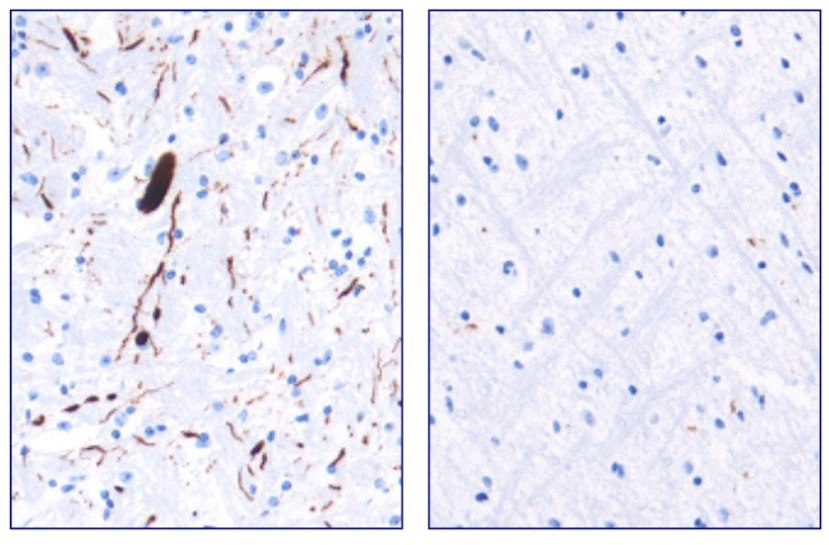
They next tested Anle138b in both cell culture and rodent models of Parkinson’s and found it to be neuroprotective and very good at inhibiting the toxic form of alpha synuclein. And the treatment looks to be very effective. In the image below you can see dark staining of toxic alpha synuclein in the left panel from the brain of an untreated mouse, but very little staining in the right panel from an Anle138b treated mouse.
Toxic form of alpha synuclein (dark staining). Source: Max-Planck
Importantly, Anle138b does not interfere with the production and normal behaviour of alpha synuclein protein in the mice, but it does act as an inhibitor of alpha synuclein aggregation (which is considered to be the toxic form of the protein). In addition, the investigators found no toxic effects of Anle138b in any of their experiments even after long-term high-dose treatment (more than one year).
And in a follow up study, the drug was effective even if it was given after the disease model had started:
Title: The oligomer modulator anle138b inhibits disease progression in a Parkinson mouse model even with treatment started after disease onset
Authors: Levin J, Schmidt F, Boehm C, Prix C, Bötzel K, Ryazanov S, Leonov A, Griesinger C, Giese A.
Journal: Acta Neuropathol. 2014 May;127(5):779-80.
PMID: 24615514 (This article is OPEN ACCESS if you would like to read it)
During the first study, the researchers had started Anle138b treatment in the mouse model of Parkinson’s at a very young age. In this study, however, the investigators used genetically engineered mice which produce high levels of a mutant version of the human alpha synuclein protein (A30-P), and began treatment only as the symptoms were starting to show. These mice begin to display Parkinson’s-like symptoms at approximately 300 days of age. The researchers began treating the mice at 350 days of age and they found that Anle138b was significantly improved the overall survival of the mice.
More recently, the researchers followed up this research by investigating Anle138b in a mouse model of MSA:

Authors: Heras-Garvin A, Weckbecker D, Ryazanov S, Leonov A, Griesinger C, Giese A, Wenning GK, Stefanova N.
Journal: Mov Disord. 2019 Feb;34(2):255-263. doi: 10.1002/mds.27562. Epub 2018 Nov 19.
PMID: 30452793 (This report is OPEN ACCESS if you would like to read it)
The PLP‐hαSyn mouse model recapitulates many of the clinical and pathophysiological features of MSA, and the investigators who conducted this study used these mice to test the potential of Anle138b as a treatment against MSA. The researchers found that treating the mice for 4 months with Anle138b significantly reduced alpha synuclein accumulation, resulting in neuroprotection and a reduction of immune system activation in the mice (compared to placebo treated animals). Anle138b treatment also helped to preserve motor function in the mice.
And these results have given MODAG confidence in moving to clinical trials of Anle138b in humans.
What about Parkinson’s? Has Anle138b been tested in model of Parkinson’s?
Yes. A research group at Cambridge university has demonstrated the potential of Anle138b in a new mouse model of Parkinson’s:

Authors: Wegrzynowicz M, Bar-On D, Calo’ L, Anichtchik O, Iovino M, Xia J, Ryazanov S, Leonov A, Giese A, Dalley JW, Griesinger C, Ashery U, Spillantini MG.
Journal: Acta Neuropathol. 2019 Oct;138(4):575-595.
PMID: 31165254
In this study, the investigators generated a new mouse model of Parkinson’s (called MI2), which produces high levels of the human alpha synuclein protein which is particularly aggregation-prone (truncated 1–120 α-syn). And they engineered these mice so that this protein was only produce in the dopamine neurons of the brain.
These mice displayed a progressive reduction of dopamine levels and significant dopamine cell death starting from 6 and 12 months of age, respectively. The animals began displaying the first signs of motor issues from about 9 months of age and gross motor probelms at 20 months of age (when 50% of dopamine neurons had been lost). This mouse model appears to resemble the human condition very well.
Remarkably, treatment with Anle138b from 9 to 12 months of age restored dopamine levels, prevented dopamine cell death and improved the motor impairments in this mice. Despite the delayed treatment, Anle138b was still able to have a beneficial effect in these mice. This finding validates and expands of similar previous studies involving delayed treatment onset with Anle138b (Click here to read more about this).
Has Anle138b been tested in humans?
Yes it has.
In August 2020, MODAG announced that they had completed their first Phase I clinical study of Anle138b in healthy volunteers (Click here to read more about this), and the results were very pleasing. Tested across different doses, the investigators found that at a 100 mg dose-level, anle138b blood levels were significantly above the threshold dose that has been found to be effective in the preclinical experiments.
In addition, the treatment was very safe, with no therapy-specific side effects being reported. The half-life of anle138b after oral administration in capsule form was approximately 12 hours, which makes it ideal for a potential once-daily application (if daily treatment is required), and the absorption of the drug was not significantly altered by food intake. Overall, very positive results.
Then December 2020, the company initiated a Phase Ib clinical trial of anle138b in 24 individuals with mild to moderate Parkinson´s. This study is being conducted by Quotient Sciences in Nottingham (UK) with support from the Neurology Department of Nottingham University Hospital. The study will assess the safety, tolerability, and pharmacokinetics different doses of anle138b for seven days (Click here to read more about this study). This U.K. study is being supported by the Michael J. Fox Foundation and is scheduled to finish in June 2021.
No.
Early on, the team at MODAG realised that the current tools for assessing target engagement and efficacy of alpha synuclein aggregation inhibitors were… well, “lacking” is not really appropriate, perhaps “non-existent” is probably more apt. So the company started working on imaging agents that would allow them to better quantify the amount of aggregated alpha synuclein in the brain before and after a person is treated with an experimental therapy, like anle138b.
And last year they published some of the research that they have been conducting in this area.

Authors: Maurer A, Leonov A, Ryazanov S, Herfert K, Kuebler L, Buss S, Schmidt F, Weckbecker D, Linder R, Bender D, Giese A, Pichler BJ, Griesinger C.
Journal: ChemMedChem. 2020 Mar 5;15(5):411-415.
PMID: 31859430 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers described a radiolabeling strategy for the aggregation modulator anle253b. They also demonstrated its high‐affinity binding to aggregated alpha synuclein in cell culture conditions and its successful penetration of the blood‐brain barrier in rats.
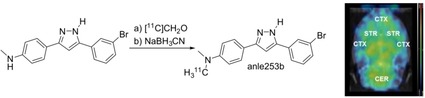
And this study was quickly followed by a second study:

Authors: Kuebler L, Buss S, Leonov A, Ryazanov S, Schmidt F, Maurer A, Weckbecker D, Landau AM, Lillethorup TP, Bleher D, Saw RS, Pichler BJ, Griesinger C, Giese A, Herfert K.
Journal: Eur J Nucl Med Mol Imaging. 2020 Dec 28. Online ahead of print.
PMID: 33369690 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers reported the development of a positron emission tomography (PET) brain imaging tracer based on anle138b, which they called [3H]MODAG-001.
The agent revealed a very high affinity towards aggregated alpha synuclein protein (and moderate affinity to aggregated Tau and beta amyloid), as well as excellent CNS penetrance in mice. It will be interesting to watch the future development of this research – an imaging agent for alpha synuclein is currently considered the holy grail for the assessment of clinical trials focused on disease modification in Parkinson’s.
Conducting this kind of readout research speaks volumes to the ethic of the organisation involved. It suggests that the team is not just focused on making money with a new wonder drug, but also on improving the research field. It also indicates a prudence that investors can appreciate – it says “we want to be certain that our agent is doing what we say it is doing“.
Ok. So what is the new deal between TEAV and MODAG all about?
According to the press release, Teva Pharmaceutical and MODAG have announced a strategic collaboration on the exclusive worldwide licensing and development of MODAG’s lead compound anle138b and a related compound, sery433.
What is sery433?
The only mention I can find of sery433 online is in a University of Göttingen doctoral thesis from early 2020. The document reads: “In order to improve the formulation protocol and potentially anle138b’s bioavailability, we designed and synthesized several water-soluble anle138b prodrugs. Among three candidates, the prodrug sery433 showed favorable physicochemical (good stability and high solubility in water) and pharmacokinetic profiles” (Click here to read more about this).
So sery433 appears to be a prodrug for Anle138b.
What is a prodrug?
A prodrug is a molecule that, after administration, is converted (or ‘metabolised’) by the body into a pharmacologically active drug. There are lots of examples of prodrugs readily used. For example, aspirin (or acetylsalicylic acid) is a prodrug. Once in the body, aspirin is metabolised into salicylic acid which is the active molecule.
Prodrugs can be a useful means of improving delivery of a treatment (if it is necessary).
What else did the press release say?
The press release said that the companies will jointly develop the compounds for the multiple system atrophy (or MSA) and Parkinson’s.
MSA – also known as Shy-Drager syndrome – is a rare neurodegenerative condition that is often misdiagnosed as Parkinson’s. It is considered an atypical Parkinsonism and it is believed to be influenced by aggregated alpha synuclein (Click here to read a previous SoPD post about MSA).
According to the press release, MODAG plans to conduct long-term clinical trials investigating the efficacy of anle138b in both MSA and Parkinson’s. “These studies are anticipated to begin at the end of 2021” (Read the press release for more information).
So what does it all mean?
Several years ago, the Parkinson’s community was concerned by the perceived exodus of big pharmaceutical companies from research on neurodegenerative conditions. This was largely associated with the Pfizer news in early 2018 (Click here to read a SoPD rant about PR disaster). While TEVA is not necessarily purchasing MODAG, it is certainly expressing a significant interest in the agent being developed. And this represents a continuing theme of the last 18 months in which other small biotech firms developing potentially disease modifying treatments for Parkinson’s are quietly being partnered up.
Some of the more prominent examples were:
- Biogen tied a knot with Denali (Click here to read an SoPD post about this)
- Bayer acquired Asklepios BioPharmaceutical which then merged with Brain Neurotherapy Bio -the GDNF gene therapy biotech firm (Click here to read a SoPD post about this – and don’t forget that Bayer purchased BlueRock Therapeutics – the stem cell transplantation company last year – click here to read a SoPD post about that).
- Bial purchased Lysosomal Therapeutics (Click here to read more about this)
- Eli Lilly acquired GBA-associated Parkinson’s gene therapy company Prevail Therapeutics (Click here to read a SoPD post on this).
- AC Immune acquires assets of AFFiRiS (Click here to read a SoPD post about this).
- Inflammasome-focused biotech firm Inflazome was bought by Roche (Click here to read more about this).
Anle138b is an experimental drug that we here at the SoPD are keeping a close eye on.
As a small molecule it is able to cross the blood-brain-barrier (the protective membrane protecting the brain from the outside world) and cross cell membranes to deal with aggregated alpha synuclein inside of neurons. This is something that antibodies-targeting alpha synuclein – a class of experimental therapeutics that are more clinically advanced in their development – are unable to do. Thus, anle138b will be one of the first real tests of the long-held theories that clustered alpha synuclein are a driving influencer of Parkinson’s progression.
We eagerly await the details of the planned long-term clinical trials investigating the efficacy of anle138b in Parkinson’s.

Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
The banner for today’s post was sourced from TEVA and MODAG.